Scientists Uncover Source of Helium-3 Leakage from Earth's Core
Helium-3 is a very rare element on Earth, but it is a very promising fuel for nuclear fusion reactors. If we can figure out how to extract it from the Earth's core, it could provide us with a clean and abundant source of energy.
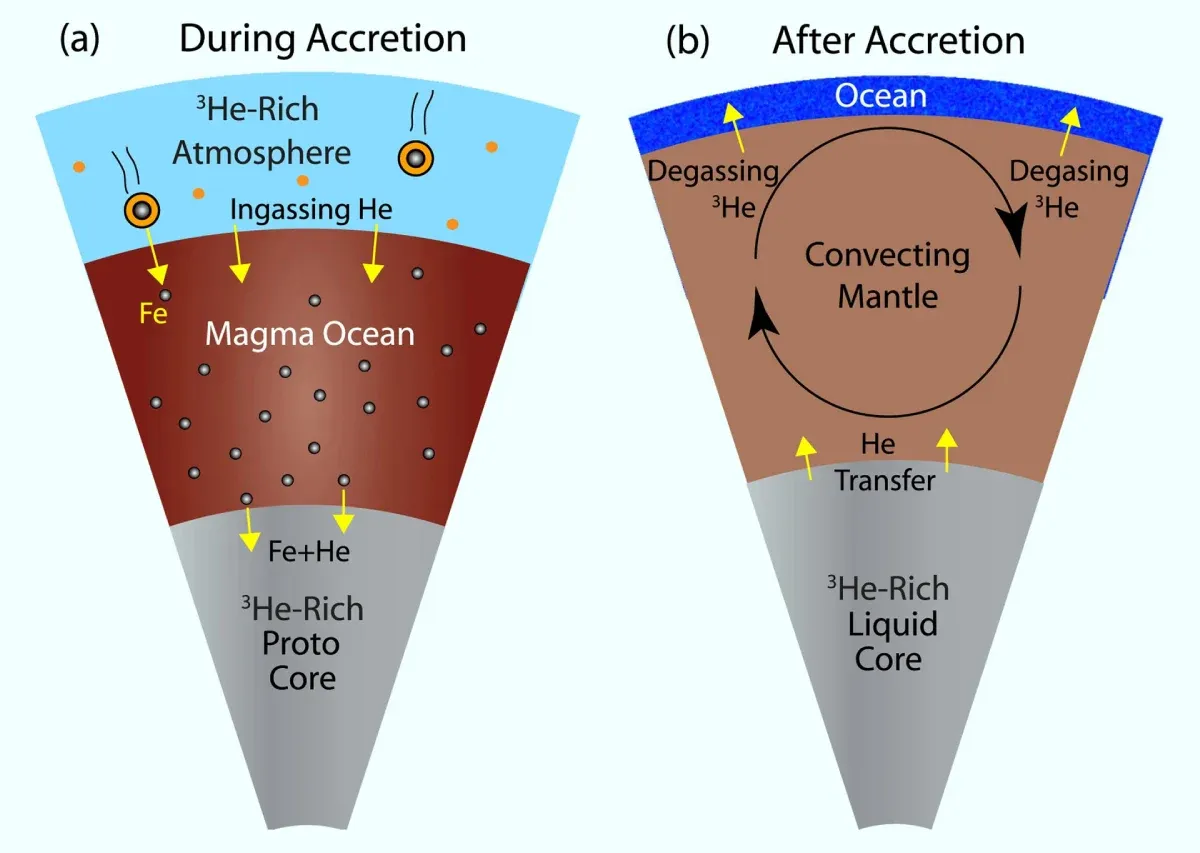
The new study suggests that helium-3 is leaking out of the Earth's core through a process called exsolution. This is when a solid mineral breaks down into two or more different minerals. In this case, the mineral magnesium oxide is breaking down into magnesium and helium-3.
Scientists think they may have solved the mystery of how helium-3, a rare element that has attracted interest as a potential fuel source for nuclear fusion reactors, is leaking out of Earth’s core, reports a new study.
The research proposes that this ancient substance seeps from the core into the planet’s outer layers, the mantle and crust, as a result of reactions with magnesium oxide. The discovery could help probe Earth’s mysterious innermost regions, and has implications for understanding how life emerged on Earth and whether it might exist elsewhere in the universe.
Helium-3, a helium isotope with one neutron in its nucleus instead of the more common two, is extremely rare on Earth's surface, but recent research has shown that it can be found in volcanic rock formations on the ocean floor. The finding suggests that some process allows helium-3 to seep from the core into the mantle. Scientists would like to obtain more helium-3 because it has proven useful in fusion reactors as a source of fuel.
Prior research has suggested that there is a large amount of helium-3 locked in Earth's core—planetary scientists believe it got there during the formation of the planet. Helium-3 was a constituent of the dust and gases that coalesced approximately 4.5 billion years ago. And because of its nature, it became locked into the core. But some process has been allowing some of it to escape into the mantle, and now, Deng and Du believe they may have figured out why.
Prior research has shown that when helium-3 comes into contact with magnesium oxide, it exsolves, which means it is changed from an initial homogenous mineral into one that has crystalline phases. As part of the process, the helium is physically moved to the location of the magnesium oxide.
The researchers mathematically demonstrated that helium-3 could have been pulled from the core through interactions with magnesium oxide shortly after the planet formed, continuing right up to the present. Their math shows that over the lifetime of the planet, as helium-3 was exsolved, it could have slowly made its way to parts of the mantle, where it may have risen to the surface through volcanic activity.
The above story is based on Materials provided by Guangzhou Institute of Geochemistry.